Seznamy 104+ Sp Hybridised Nitrogen Atom Vynikající
Seznamy 104+ Sp Hybridised Nitrogen Atom Vynikající. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z).
Nejlepší Hybrid Orbitals Chemistry Libretexts
N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. If the steric number is 4, it is sp3. The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. In ethene molecule, each carbon atom undergoes sp 2 hybridisation. In fact, there is sp3 hybridization on each nitrogen.So now, let's go back to our molecule and determine the hybridization states for all the atoms.
Two sp orbitals will be at 180 degrees to each other. Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. Only in hcn nitrogen atom is sp hybridised. Now if we consider the nitrogen atom in the pyridine, it is sp2 hybridized. In ethene molecule, each carbon atom undergoes sp 2 hybridisation. Two sp orbitals will be at 180 degrees to each other.

Both of these atoms are sp hybridized. Learn chemistry with sunil warhadpande So now, let's go back to our molecule and determine the hybridization states for all the atoms. The global maximum barrier torsional energy (when the lone pair eclipses the … 21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair.. One sp 2 hybrid orbital of one carbon atom overlaps axially.

When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons).. The global maximum barrier torsional energy (when the lone pair eclipses the … Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. In sp hybridization, the s orbital overlaps with only one p orbital. One sp 2 hybrid orbital of one carbon atom overlaps axially. Two sp orbitals will be at 180 degrees to each other. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be:. So now, let's go back to our molecule and determine the hybridization states for all the atoms.

In fact, there is sp3 hybridization on each nitrogen... However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. Carbon has 1 sigma bond each to h and n. If the steric number is 4, it is sp3. One sp 2 hybrid orbital of one carbon atom overlaps axially. In ethene molecule, each carbon atom undergoes sp 2 hybridisation... Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation.
.jpg?revision=1&size=bestfit&width=601&height=192)
21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair.. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: Now if we consider the nitrogen atom in the pyridine, it is sp2 hybridized. Only in hcn nitrogen atom is sp hybridised. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; If the steric number is 4, it is sp3.
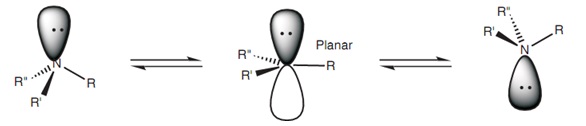
One sp 2 hybrid orbital of one carbon atom overlaps axially... Only in hcn nitrogen atom is sp hybridised. Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. If the steric number is 4, it is sp3. Two sp orbitals will be at 180 degrees to each other. Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. Learn chemistry with sunil warhadpande The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. The global maximum barrier torsional energy (when the lone pair eclipses the …. One sp 2 hybrid orbital of one carbon atom overlaps axially.

Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation.. In ethene molecule, each carbon atom undergoes sp 2 hybridisation. Both of these atoms are sp hybridized.. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons).

Only in hcn nitrogen atom is sp hybridised. If the steric number is 4, it is sp3. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). One sp 2 hybrid orbital of one carbon atom overlaps axially... When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons).

Both of these atoms are sp hybridized. If the steric number is 4, it is sp3.
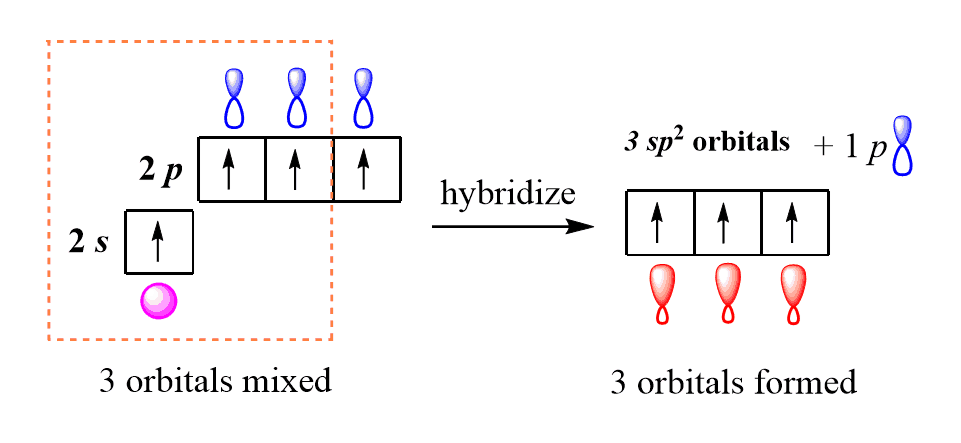
In fact, there is sp3 hybridization on each nitrogen. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). So now, let's go back to our molecule and determine the hybridization states for all the atoms... Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation.

The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3.

Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. So now, let's go back to our molecule and determine the hybridization states for all the atoms. In fact, there is sp3 hybridization on each nitrogen. In ethene molecule, each carbon atom undergoes sp 2 hybridisation. Only in hcn nitrogen atom is sp hybridised. Carbon has 1 sigma bond each to h and n. Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. The global maximum barrier torsional energy (when the lone pair eclipses the … One sp 2 hybrid orbital of one carbon atom overlaps axially.. If the steric number is 4, it is sp3.

25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). One sp 2 hybrid orbital of one carbon atom overlaps axially. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). Two sp orbitals will be at 180 degrees to each other. Only in hcn nitrogen atom is sp hybridised.. Only in hcn nitrogen atom is sp hybridised.

When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). In ethene molecule, each carbon atom undergoes sp 2 hybridisation. Carbon has 1 sigma bond each to h and n. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). So now, let's go back to our molecule and determine the hybridization states for all the atoms. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z)... In sp hybridization, the s orbital overlaps with only one p orbital.

In fact, there is sp3 hybridization on each nitrogen. Carbon has 1 sigma bond each to h and n.. In sp hybridization, the s orbital overlaps with only one p orbital.

The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals... Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. So now, let's go back to our molecule and determine the hybridization states for all the atoms. Carbon has 1 sigma bond each to h and n. Learn chemistry with sunil warhadpande Now if we consider the nitrogen atom in the pyridine, it is sp2 hybridized. If the steric number is 4, it is sp3. The global maximum barrier torsional energy (when the lone pair eclipses the … 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. In sp hybridization, the s orbital overlaps with only one p orbital. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair.

Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation.. Only in hcn nitrogen atom is sp hybridised. Both of these atoms are sp hybridized. Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). Learn chemistry with sunil warhadpande. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair.

One sp 2 hybrid orbital of one carbon atom overlaps axially.. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be:.. Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented;

When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). In sp hybridization, the s orbital overlaps with only one p orbital. Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). The global maximum barrier torsional energy (when the lone pair eclipses the … Learn chemistry with sunil warhadpande The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair... Two sp orbitals will be at 180 degrees to each other.

Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. So now, let's go back to our molecule and determine the hybridization states for all the atoms. Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; Now if we consider the nitrogen atom in the pyridine, it is sp2 hybridized. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals.. Now if we consider the nitrogen atom in the pyridine, it is sp2 hybridized.

Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. In sp hybridization, the s orbital overlaps with only one p orbital. The global maximum barrier torsional energy (when the lone pair eclipses the … Two sp orbitals will be at 180 degrees to each other. Now if we consider the nitrogen atom in the pyridine, it is sp2 hybridized. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. The global maximum barrier torsional energy (when the lone pair eclipses the …

Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma).. The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals.

When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons).. Both of these atoms are sp hybridized... However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z).

The global maximum barrier torsional energy (when the lone pair eclipses the … 21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be:. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be:

Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be:. Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented;
In ethene molecule, each carbon atom undergoes sp 2 hybridisation. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. Learn chemistry with sunil warhadpande In sp hybridization, the s orbital overlaps with only one p orbital. One sp 2 hybrid orbital of one carbon atom overlaps axially. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). Only in hcn nitrogen atom is sp hybridised. Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. If the steric number is 4, it is sp3. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma).

25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: Only in hcn nitrogen atom is sp hybridised. Now if we consider the nitrogen atom in the pyridine, it is sp2 hybridized. If the steric number is 4, it is sp3.
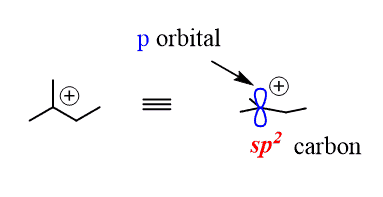
N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. One sp 2 hybrid orbital of one carbon atom overlaps axially. Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; In fact, there is sp3 hybridization on each nitrogen. The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. In ethene molecule, each carbon atom undergoes sp 2 hybridisation. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: So now, let's go back to our molecule and determine the hybridization states for all the atoms.. Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation.

When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons)... 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. 21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair. One sp 2 hybrid orbital of one carbon atom overlaps axially. In fact, there is sp3 hybridization on each nitrogen. In sp hybridization, the s orbital overlaps with only one p orbital.. Both of these atoms are sp hybridized.

In ethene molecule, each carbon atom undergoes sp 2 hybridisation.. If the steric number is 4, it is sp3. Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented;

N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. Now if we consider the nitrogen atom in the pyridine, it is sp2 hybridized. Carbon has 1 sigma bond each to h and n. Two sp orbitals will be at 180 degrees to each other. Both of these atoms are sp hybridized. Only in hcn nitrogen atom is sp hybridised. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. The global maximum barrier torsional energy (when the lone pair eclipses the … N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. In sp hybridization, the s orbital overlaps with only one p orbital.

N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. 21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). In fact, there is sp3 hybridization on each nitrogen. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; In sp hybridization, the s orbital overlaps with only one p orbital. So now, let's go back to our molecule and determine the hybridization states for all the atoms. If the steric number is 4, it is sp3.. In ethene molecule, each carbon atom undergoes sp 2 hybridisation.

Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. So now, let's go back to our molecule and determine the hybridization states for all the atoms. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). Now if we consider the nitrogen atom in the pyridine, it is sp2 hybridized. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). Two sp orbitals will be at 180 degrees to each other. The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. Carbon has 1 sigma bond each to h and n.

Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation... So now, let's go back to our molecule and determine the hybridization states for all the atoms. Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. Two sp orbitals will be at 180 degrees to each other. Learn chemistry with sunil warhadpande When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). Only in hcn nitrogen atom is sp hybridised.. The global maximum barrier torsional energy (when the lone pair eclipses the …

Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation.. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). In sp hybridization, the s orbital overlaps with only one p orbital. Carbon has 1 sigma bond each to h and n. Only in hcn nitrogen atom is sp hybridised. In fact, there is sp3 hybridization on each nitrogen... So now, let's go back to our molecule and determine the hybridization states for all the atoms.
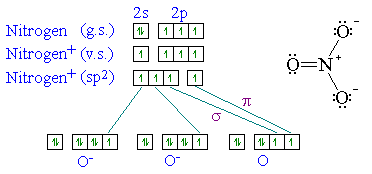
When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. In ethene molecule, each carbon atom undergoes sp 2 hybridisation. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. Two sp orbitals will be at 180 degrees to each other.. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z).

So now, let's go back to our molecule and determine the hybridization states for all the atoms. 21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair. Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. In ethene molecule, each carbon atom undergoes sp 2 hybridisation. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). So now, let's go back to our molecule and determine the hybridization states for all the atoms. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). Only in hcn nitrogen atom is sp hybridised. One sp 2 hybrid orbital of one carbon atom overlaps axially.. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3.

So now, let's go back to our molecule and determine the hybridization states for all the atoms. Two sp orbitals will be at 180 degrees to each other. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation.. Learn chemistry with sunil warhadpande

Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization.. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. 21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). So now, let's go back to our molecule and determine the hybridization states for all the atoms.

The global maximum barrier torsional energy (when the lone pair eclipses the …. Only in hcn nitrogen atom is sp hybridised. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). Carbon has 1 sigma bond each to h and n. Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. 21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair. Now if we consider the nitrogen atom in the pyridine, it is sp2 hybridized. Two sp orbitals will be at 180 degrees to each other. In fact, there is sp3 hybridization on each nitrogen. Both of these atoms are sp hybridized. Learn chemistry with sunil warhadpande The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals.

One sp 2 hybrid orbital of one carbon atom overlaps axially... Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). In ethene molecule, each carbon atom undergoes sp 2 hybridisation. If the steric number is 4, it is sp3. In sp hybridization, the s orbital overlaps with only one p orbital. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). 21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair. Learn chemistry with sunil warhadpande. Both of these atoms are sp hybridized.
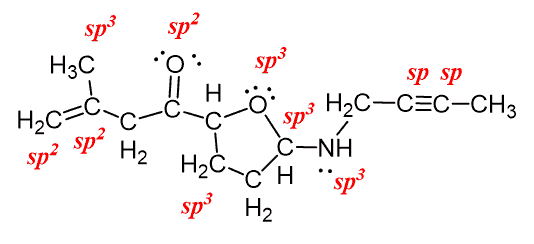
Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented;.. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. In ethene molecule, each carbon atom undergoes sp 2 hybridisation. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). Only in hcn nitrogen atom is sp hybridised.. If the steric number is 4, it is sp3.

In sp hybridization, the s orbital overlaps with only one p orbital... 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. If the steric number is 4, it is sp3. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). The global maximum barrier torsional energy (when the lone pair eclipses the … N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: In ethene molecule, each carbon atom undergoes sp 2 hybridisation. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z).. In fact, there is sp3 hybridization on each nitrogen.

If the steric number is 4, it is sp3... Carbon has 1 sigma bond each to h and n. Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3... Both of these atoms are sp hybridized.

In fact, there is sp3 hybridization on each nitrogen. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). In fact, there is sp3 hybridization on each nitrogen. Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation. The global maximum barrier torsional energy (when the lone pair eclipses the … Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. If the steric number is 4, it is sp3. In sp hybridization, the s orbital overlaps with only one p orbital.
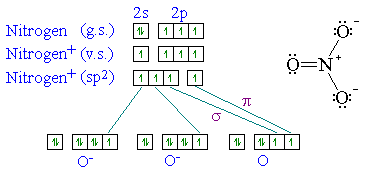
So now, let's go back to our molecule and determine the hybridization states for all the atoms.. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). In fact, there is sp3 hybridization on each nitrogen. Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. The global maximum barrier torsional energy (when the lone pair eclipses the … Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. 21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair. If the steric number is 4, it is sp3. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. Carbon has 1 sigma bond each to h and n... Two sp orbitals will be at 180 degrees to each other.

Carbon has 1 sigma bond each to h and n. Two sp orbitals will be at 180 degrees to each other.. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z).

If the steric number is 4, it is sp3. In ethene molecule, each carbon atom undergoes sp 2 hybridisation. So now, let's go back to our molecule and determine the hybridization states for all the atoms. If the steric number is 4, it is sp3. In fact, there is sp3 hybridization on each nitrogen. Carbon has 1 sigma bond each to h and n. Both of these atoms are sp hybridized. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. Only in hcn nitrogen atom is sp hybridised. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair.

25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: If the steric number is 4, it is sp3. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). In sp hybridization, the s orbital overlaps with only one p orbital. The global maximum barrier torsional energy (when the lone pair eclipses the … One sp 2 hybrid orbital of one carbon atom overlaps axially. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: So now, let's go back to our molecule and determine the hybridization states for all the atoms. In ethene molecule, each carbon atom undergoes sp 2 hybridisation. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons).

Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; Both of these atoms are sp hybridized. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). If the steric number is 4, it is sp3. Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. 21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair. One sp 2 hybrid orbital of one carbon atom overlaps axially. In sp hybridization, the s orbital overlaps with only one p orbital.

Only in hcn nitrogen atom is sp hybridised. If the steric number is 4, it is sp3. Learn chemistry with sunil warhadpande However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z). In fact, there is sp3 hybridization on each nitrogen. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). So now, let's go back to our molecule and determine the hybridization states for all the atoms. Learn chemistry with sunil warhadpande
Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; So now, let's go back to our molecule and determine the hybridization states for all the atoms. In sp hybridization, the s orbital overlaps with only one p orbital.

One sp 2 hybrid orbital of one carbon atom overlaps axially. The global maximum barrier torsional energy (when the lone pair eclipses the … Carbon has 1 sigma bond each to h and n. Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented; Both of these atoms are sp hybridized. One sp 2 hybrid orbital of one carbon atom overlaps axially. In ethene molecule, each carbon atom undergoes sp 2 hybridisation. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). In fact, there is sp3 hybridization on each nitrogen... In fact, there is sp3 hybridization on each nitrogen.

21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair.. Now if we consider the nitrogen atom in the pyridine, it is sp2 hybridized. One sp 2 hybrid orbital of one carbon atom overlaps axially. In fact, there is sp3 hybridization on each nitrogen. Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3. Both of these atoms are sp hybridized. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. In sp hybridization, the s orbital overlaps with only one p orbital. So now, let's go back to our molecule and determine the hybridization states for all the atoms. Only in hcn nitrogen atom is sp hybridised. In ethene molecule, each carbon atom undergoes sp 2 hybridisation.

21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair... Two sp orbitals will be at 180 degrees to each other. So now, let's go back to our molecule and determine the hybridization states for all the atoms. Only in hcn nitrogen atom is sp hybridised. 06.05.2015 · (i will prove this point indirectly further using an experimental data), the nitrogen atom hybridization is a linear combination of sp2 and sp3.. However, following hunds rule, the 3 p electrons occupy 3 degenerate p orbitals (x, y and z).

In sp hybridization, the s orbital overlaps with only one p orbital... In ethene molecule, each carbon atom undergoes sp 2 hybridisation.

The unhybridized 2p1 orbital lies perpendicular to the three hybridised orbitals. Two sp orbitals will be at 180 degrees to each other. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). Both of these atoms are sp hybridized. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: In fact, there is sp3 hybridization on each nitrogen. So now, let's go back to our molecule and determine the hybridization states for all the atoms... Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented;

The global maximum barrier torsional energy (when the lone pair eclipses the ….. If the steric number is 4, it is sp3. Carbon has 1 sigma bond each to h and n. When the s and p orbitals are hybridised to create sp3 hybrid orbitals, we have 4 hybrid orbitals to be filled with 5 electrons (because nitrogen has 5 valence electrons). Learn chemistry with sunil warhadpande One sp 2 hybrid orbital of one carbon atom overlaps axially. The global maximum barrier torsional energy (when the lone pair eclipses the … 21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair.. Learn chemistry with sunil warhadpande

21.09.2021 · notice that, while carbon also has a single bond to hydrogen, the nitrogen has no other bond, just a lone pair. One sp 2 hybrid orbital of one carbon atom overlaps axially. In sp hybridization, the s orbital overlaps with only one p orbital. 25.07.2016 · nitrogen has atomic number 7, so electronic configuration will be: Representation of sp 2 hybridization sp 2 hybridization is also known as trigonal hybridisation.. Carbon has 1 sigma bond each to h and n.