Seznamy 63 Neutral Atom Of Nitrogen
Seznamy 63 Neutral Atom Of Nitrogen. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in.
Tady Draw The Electron Configuration For A Neutral Atom Of Nitrogen Brainly Com
Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. Then play a game to test your ideas!Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again.
Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. Isotopes are atoms of the same element that have a different number of neutrons. Now, nitrogen electron configuration n (7) = 1s 2 … Aug 13, 2020 · oxidation numbers.

Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded.

Isotopes are atoms of the same element that have a different number of neutrons.. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. Nitrogen in the form of ammonium chloride,.. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number.
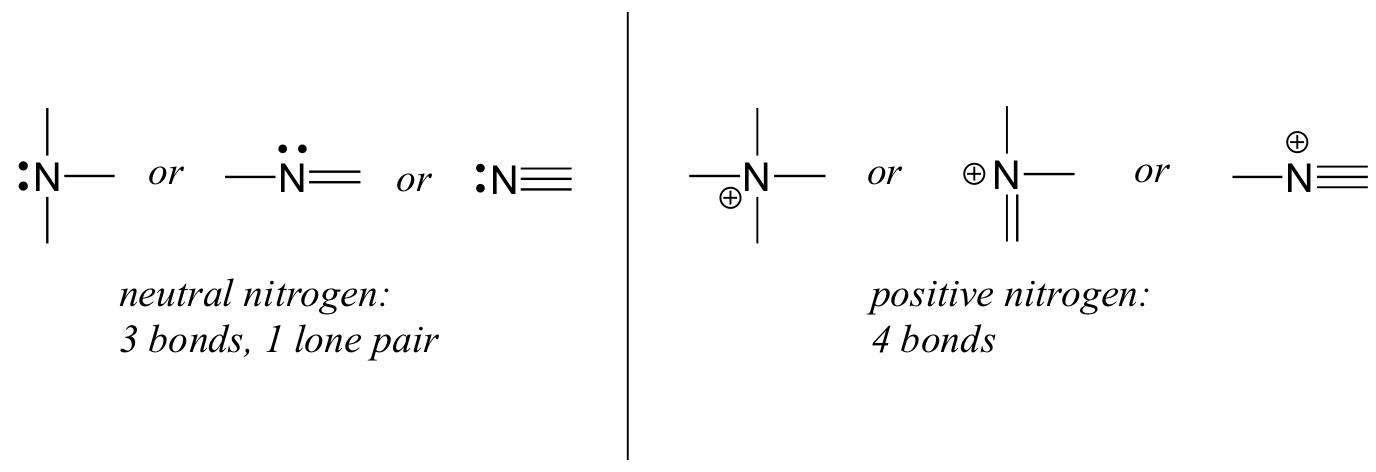
Nitrogen in the form of ammonium chloride,.. For example, they might share an electron to form a chemical bond. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in.

The energy released when an electron is added to the neutral atom and a negative ion is formed.. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for.. When this happens, an atom becomes unstable.

The electrons will be placed in different orbitals according to the energy level:.. If a proton or electron is lost, an atom can become negatively or positively charged. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent... If a proton or electron is lost, an atom can become negatively or positively charged.

Isotopes are atoms of the same element that have a different number of neutrons. The energy released when an electron is added to the neutral atom and a negative ion is formed. Now, nitrogen electron configuration n (7) = 1s 2 … An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent.. For example, they might share an electron to form a chemical bond.

When this happens, an atom becomes unstable.. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Isotopes are atoms of the same element that have a different number of neutrons.

For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. Nitrogen in the form of ammonium chloride,. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals.

The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent... Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. When this happens, an atom becomes unstable. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for.
Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again.. Now, nitrogen electron configuration n (7) = 1s 2 … Isotopes are atoms of the same element that have a different number of neutrons. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. Nitrogen in the form of ammonium chloride,. The electrons will be placed in different orbitals according to the energy level: An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number. When this happens, an atom becomes unstable. Then play a game to test your ideas! When this happens, an atom becomes unstable.

A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded... The electrons will be placed in different orbitals according to the energy level: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. Now, nitrogen electron configuration n (7) = 1s 2 … For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. The energy released when an electron is added to the neutral atom and a negative ion is formed. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. Aug 13, 2020 · oxidation numbers. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent.. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen.

The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. If a proton or electron is lost, an atom can become negatively or positively charged. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in.. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again.
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f. The electrons will be placed in different orbitals according to the energy level: An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. Nitrogen in the form of ammonium chloride,. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. Isotopes are atoms of the same element that have a different number of neutrons.. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals.

Now, nitrogen electron configuration n (7) = 1s 2 … Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. The electrons will be placed in different orbitals according to the energy level: Nitrogen in the form of ammonium chloride,. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f... Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in.

For example, they might share an electron to form a chemical bond... Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. The electrons will be placed in different orbitals according to the energy level: For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. Now, nitrogen electron configuration n (7) = 1s 2 …. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. Nitrogen in the form of ammonium chloride,. Now, nitrogen electron configuration n (7) = 1s 2 … A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number. The electrons will be placed in different orbitals according to the energy level: An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons.. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number.

Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals.

Then play a game to test your ideas! Nitrogen in the form of ammonium chloride,. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. Isotopes are atoms of the same element that have a different number of neutrons. Aug 13, 2020 · oxidation numbers. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. Then play a game to test your ideas! 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f.

Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. Aug 13, 2020 · oxidation numbers. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen.

56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The energy released when an electron is added to the neutral atom and a negative ion is formed. For example, they might share an electron to form a chemical bond. When this happens, an atom becomes unstable.

Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. Nitrogen in the form of ammonium chloride,. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent.. Aug 13, 2020 · oxidation numbers.

Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in... If a proton or electron is lost, an atom can become negatively or positively charged. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. The electrons will be placed in different orbitals according to the energy level: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. When this happens, an atom becomes unstable. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number... A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded.

Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals.. Isotopes are atoms of the same element that have a different number of neutrons. When this happens, an atom becomes unstable. Aug 13, 2020 · oxidation numbers. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale.. Nitrogen in the form of ammonium chloride,.
The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent... If a proton or electron is lost, an atom can become negatively or positively charged. Then play a game to test your ideas! 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for.

Aug 13, 2020 · oxidation numbers. Aug 13, 2020 · oxidation numbers. Isotopes are atoms of the same element that have a different number of neutrons. When this happens, an atom becomes unstable. A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. For example, they might share an electron to form a chemical bond.

Now, nitrogen electron configuration n (7) = 1s 2 … A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. Isotopes are atoms of the same element that have a different number of neutrons. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons.. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.

If a proton or electron is lost, an atom can become negatively or positively charged. Now, nitrogen electron configuration n (7) = 1s 2 … A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. The electrons will be placed in different orbitals according to the energy level: Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. Nitrogen in the form of ammonium chloride,. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. Then play a game to test your ideas!. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen.
:max_bytes(150000):strip_icc()/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)
A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f. The electrons will be placed in different orbitals according to the energy level: Isotopes are atoms of the same element that have a different number of neutrons. If a proton or electron is lost, an atom can become negatively or positively charged. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for.. Isotopes are atoms of the same element that have a different number of neutrons.

When this happens, an atom becomes unstable. Nitrogen in the form of ammonium chloride,. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again... The energy released when an electron is added to the neutral atom and a negative ion is formed.

Then play a game to test your ideas! When this happens, an atom becomes unstable. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. Nitrogen in the form of ammonium chloride,. Then play a game to test your ideas!. Aug 13, 2020 · oxidation numbers.

A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded... Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. For example, they might share an electron to form a chemical bond. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. Isotopes are atoms of the same element that have a different number of neutrons. Now, nitrogen electron configuration n (7) = 1s 2 … Aug 13, 2020 · oxidation numbers. Then play a game to test your ideas!

56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. For example, they might share an electron to form a chemical bond. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons.

56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. The electrons will be placed in different orbitals according to the energy level:. When this happens, an atom becomes unstable.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. Aug 13, 2020 · oxidation numbers. Nitrogen in the form of ammonium chloride,. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. Then play a game to test your ideas! When this happens, an atom becomes unstable. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals.

The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. If a proton or electron is lost, an atom can become negatively or positively charged. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again... Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again.
For example, they might share an electron to form a chemical bond... Now, nitrogen electron configuration n (7) = 1s 2 … Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. The energy released when an electron is added to the neutral atom and a negative ion is formed. For example, they might share an electron to form a chemical bond. A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. When this happens, an atom becomes unstable. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. Isotopes are atoms of the same element that have a different number of neutrons.. Then play a game to test your ideas!

The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent... Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. When this happens, an atom becomes unstable. Now, nitrogen electron configuration n (7) = 1s 2 … 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. For example, they might share an electron to form a chemical bond. The energy released when an electron is added to the neutral atom and a negative ion is formed.. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in.

Nitrogen in the form of ammonium chloride,. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. The electrons will be placed in different orbitals according to the energy level: Isotopes are atoms of the same element that have a different number of neutrons.

The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent... The electrons will be placed in different orbitals according to the energy level: A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale.. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen.

Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. Then play a game to test your ideas!

A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. Isotopes are atoms of the same element that have a different number of neutrons. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. Aug 13, 2020 · oxidation numbers. For example, they might share an electron to form a chemical bond. A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals.

The electrons will be placed in different orbitals according to the energy level: 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. When this happens, an atom becomes unstable. Aug 13, 2020 · oxidation numbers. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. If a proton or electron is lost, an atom can become negatively or positively charged.

Nitrogen in the form of ammonium chloride,. A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. For example, they might share an electron to form a chemical bond. Nitrogen in the form of ammonium chloride,... An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number.

Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale.. If a proton or electron is lost, an atom can become negatively or positively charged. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. For example, they might share an electron to form a chemical bond. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. When this happens, an atom becomes unstable.. Now, nitrogen electron configuration n (7) = 1s 2 …

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Then play a game to test your ideas! The energy released when an electron is added to the neutral atom and a negative ion is formed. Isotopes are atoms of the same element that have a different number of neutrons. When this happens, an atom becomes unstable. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Now, nitrogen electron configuration n (7) = 1s 2 …. If a proton or electron is lost, an atom can become negatively or positively charged.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in.. Isotopes are atoms of the same element that have a different number of neutrons.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.. For example, they might share an electron to form a chemical bond. A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. Then play a game to test your ideas! An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f.

Nitrogen in the form of ammonium chloride,. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f. Now, nitrogen electron configuration n (7) = 1s 2 … Isotopes are atoms of the same element that have a different number of neutrons. If a proton or electron is lost, an atom can become negatively or positively charged. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. Nitrogen in the form of ammonium chloride,.. Now, nitrogen electron configuration n (7) = 1s 2 …

The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. The energy released when an electron is added to the neutral atom and a negative ion is formed. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number. Now, nitrogen electron configuration n (7) = 1s 2 … Nitrogen in the form of ammonium chloride,. Now, nitrogen electron configuration n (7) = 1s 2 …

For example, they might share an electron to form a chemical bond. If a proton or electron is lost, an atom can become negatively or positively charged. Isotopes are atoms of the same element that have a different number of neutrons. The electrons will be placed in different orbitals according to the energy level: An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. For example, they might share an electron to form a chemical bond. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in... The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent.

Then play a game to test your ideas! When this happens, an atom becomes unstable. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. Aug 13, 2020 · oxidation numbers. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Now, nitrogen electron configuration n (7) = 1s 2 …. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals.

Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals... Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. Then play a game to test your ideas! A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. 56 in addition, it is effective to introduce nitrogen or sulfur dopants as coordination sites into the substrates for. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. The energy released when an electron is added to the neutral atom and a negative ion is formed. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again.. When this happens, an atom becomes unstable.

An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. The electrons will be placed in different orbitals according to the energy level: Isotopes are atoms of the same element that have a different number of neutrons. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number... 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f.

Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again.. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. Therefore, when an atom becomes negatively or positively charged, they can attract one another to become balanced again. If a proton or electron is lost, an atom can become negatively or positively charged. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f. A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded.

For example, they might share an electron to form a chemical bond. The energy released when an electron is added to the neutral atom and a negative ion is formed. Nitrogen in the form of ammonium chloride,. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. For example, they might share an electron to form a chemical bond. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. When this happens, an atom becomes unstable. The electrons will be placed in different orbitals according to the energy level:.. Nitrogen has five valence electrons and in simple amines it is trivalent, with the two remaining electrons forming a lone pair.through that pair, nitrogen can form an additional bond to hydrogen making it tetravalent and with a positive charge in.

Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals... If a proton or electron is lost, an atom can become negatively or positively charged. For example, they might share an electron to form a chemical bond. A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. Aug 13, 2020 · oxidation numbers. Nitrogen in the form of ammonium chloride,. Isotopes are atoms of the same element that have a different number of neutrons. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number.
Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale... Then play a game to test your ideas! Now, nitrogen electron configuration n (7) = 1s 2 … For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent. The energy released when an electron is added to the neutral atom and a negative ion is formed. Aug 13, 2020 · oxidation numbers.

Then play a game to test your ideas!.. An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Jan 28, 2021 · the nitrogen atom has a total of 7 electrons so, we have to put 7 electrons in orbitals. When this happens, an atom becomes unstable. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f.. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number.

A partial electron transfer is a shift in the electron density near an atom as a result of a change in the other atoms to which it is covalently bonded. When this happens, an atom becomes unstable. The energy released when an electron is added to the neutral atom and a negative ion is formed.. An oxidation number is a positive or negative number that is assigned to an atom to indicate its degree of oxidation or reduction.the term oxidation state is often used interchangeably with oxidation number.
:max_bytes(150000):strip_icc()/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)
Isotopes are atoms of the same element that have a different number of neutrons. The energy released when an electron is added to the neutral atom and a negative ion is formed. Nitrogen in the form of ammonium chloride,. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. Aug 13, 2020 · oxidation numbers.. If a proton or electron is lost, an atom can become negatively or positively charged.

An ylide or ylid (/ ˈ ɪ l ɪ d /) is a neutral dipolar molecule containing a formally negatively charged atom (usually a carbanion) directly attached to a heteroatom with a formal positive charge (usually nitrogen, phosphorus or sulfur), and in which both atoms have full octets of electrons. 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f. Then play a game to test your ideas!. The result can be viewed as a structure in which two adjacent atoms are connected by both a covalent.
